Targeted Alpha Therapy: A Revolutionary Approach to Treating Aggressive Skin Cancer
Understanding Metastatic Melanoma
Metastatic melanoma, otherwise known as stage IV melanoma, represents one of the most aggressive and challenging forms of skin cancer. This condition is characterized by its spread from the original site to other parts of the body, making it exceptionally difficult to treat. While current therapeutic approaches, such as immunotherapy and targeted medication, provide certain benefits, their effectiveness remains limited for this aggressive cancer type.
Limitations of Conventional Treatments
Radiotherapy has emerged as a promising method in the treatment of melanoma. However, traditional beta-emitting radionuclide therapies present certain limitations. These limitations mainly arise from the low energy transfer and longer radiation range, which often result in unwanted exposure and damage to healthy tissues surrounding the tumor. A breakthrough approach is needed to minimize these off-target effects and enhance the specificity of radiotherapy for metastatic melanoma.
Introducing Targeted Alpha Therapy
In an innovative development led by a team of researchers from Japan, Assistant Professor Hiroyuki Suzuki of Chiba University, along with other experts from various esteemed institutions, targeted alpha therapy (TAT) has emerged as a potential breakthrough in the treatment of refractory skin cancer. This novel approach was recently detailed in a publication in the European Journal of Nuclear Medicine and Molecular Imaging, highlighting its promise to deliver more effective and targeted treatment for metastatic melanoma.
The Science Behind TAT
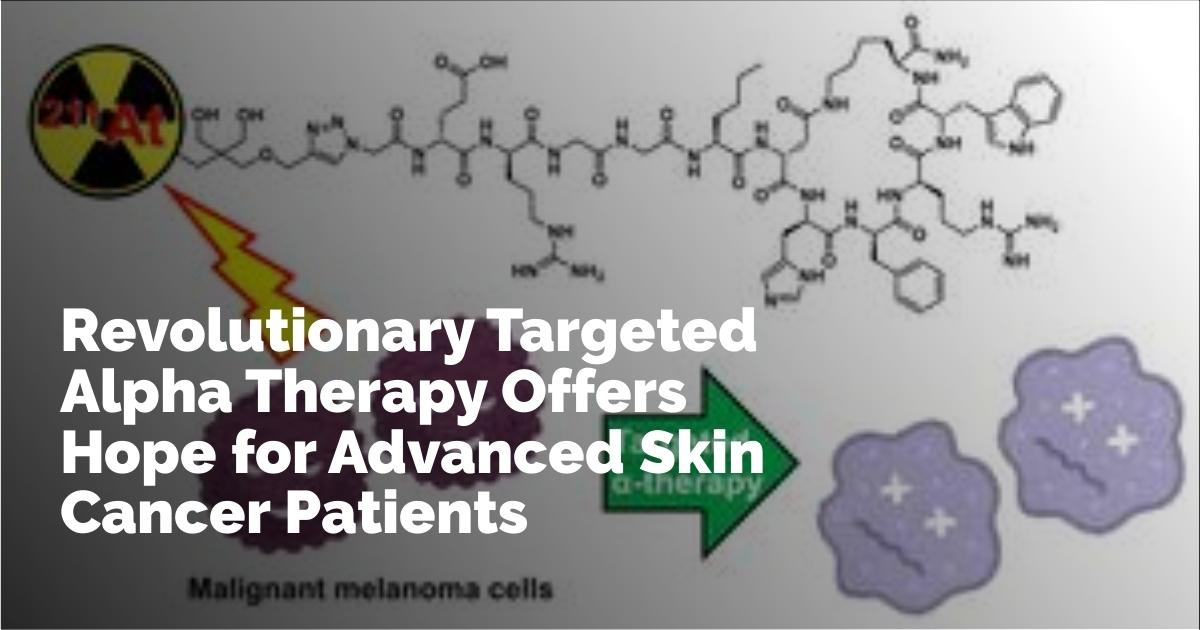
Targeted alpha therapy employs drugs that are labeled with alpha-emitting radioisotopes, specifically astatine-211 (211At), designed to target and eradicate cancer cells with precision. Alpha particles, recognized for their greater mass compared to beta and gamma emissions, offer a shorter radiation range but carry higher energy. This quality makes them particularly effective in disrupting and destroying cancer cells while sparing the surrounding healthy tissue.
Developing the Astatine-211 Labeled Drug
The innovative drug development process began with the identification of a suitable hydrophilic linker to enhance tumor specificity and reduce off-target interactions. The team developed an astatine-211 (211At)-labeled α-melanocyte-stimulating hormone (α-MSH) peptide analog, termed [211At]NpG-GGN4c. This compound specifically targets melanocortin-1 receptors (MC1R), which are prevalently overexpressed in melanoma cells, thus ensuring high tumor selectivity and minimal exposure to adjoining tissues.
Experimental Validation in Mouse Models
In order to establish the efficacy of their approach, the researchers conducted thorough testing using B16F10 melanoma-bearing mouse models. A comprehensive biodistribution analysis was then undertaken to assess tumor uptake, clearance from other organs, and general stability of the drug compound. Dr. Tomoya Uehara from Chiba University describes their methodology: “We treated the mice with varying doses of the compound, tracking tumor response, body weight, and survival rates. This allowed us to observe a dose-dependent inhibitory effect, which confirmed our approach’s effectiveness.”
Promising Results and Significant Findings
The research findings were remarkable, showcasing that the [211At]NpG-GGN4c demonstrated high accumulation in tumors with rapid clearance from non-target organs—a confirmation of its specificity for MC1R on melanoma cells. Tumor growth monitoring further revealed substantial tumor suppression, which was notably dose-dependent. The drug also exhibited high stability in blood plasma, mitigating potential risks of radioactive leakage within the body.
The Future of Refractory Cancer Treatment
Dr. Hiroyuki Suzuki expresses optimism about the potential applications of their synthesized drug’s molecular design. He notes, “Our approach holds significant promise for the development of other 211At-labeled radiopharmaceuticals, potentially opening new avenues for treating refractory cancers beyond melanoma.”
The research team is also eager to advance towards clinical application of 211At-based TAT. “If successfully translated into human trials, this therapy could emerge as a viable treatment for patients with advanced melanoma within the coming years,” anticipates Dr. Suzuki. “It could indeed offer new therapeutic opportunities for patients with refractory cancer, ushering in a new era of targeted cancer treatments.”
Conclusion
The development of targeted alpha therapy represents a monumental step forward in the treatment of metastatic melanoma, addressing the critical need for more targeted and effective treatment options. With the promise of reduced off-target effects and enhanced specificity, TAT could well redefine radiotherapy in the oncology field. As research progresses towards human clinical trials, there is hope that this advancements could revolutionize the therapeutic landscape for aggressive and treatment-resistant skin cancers, providing new hope for patients and clinicians alike.
출처 : Original Source